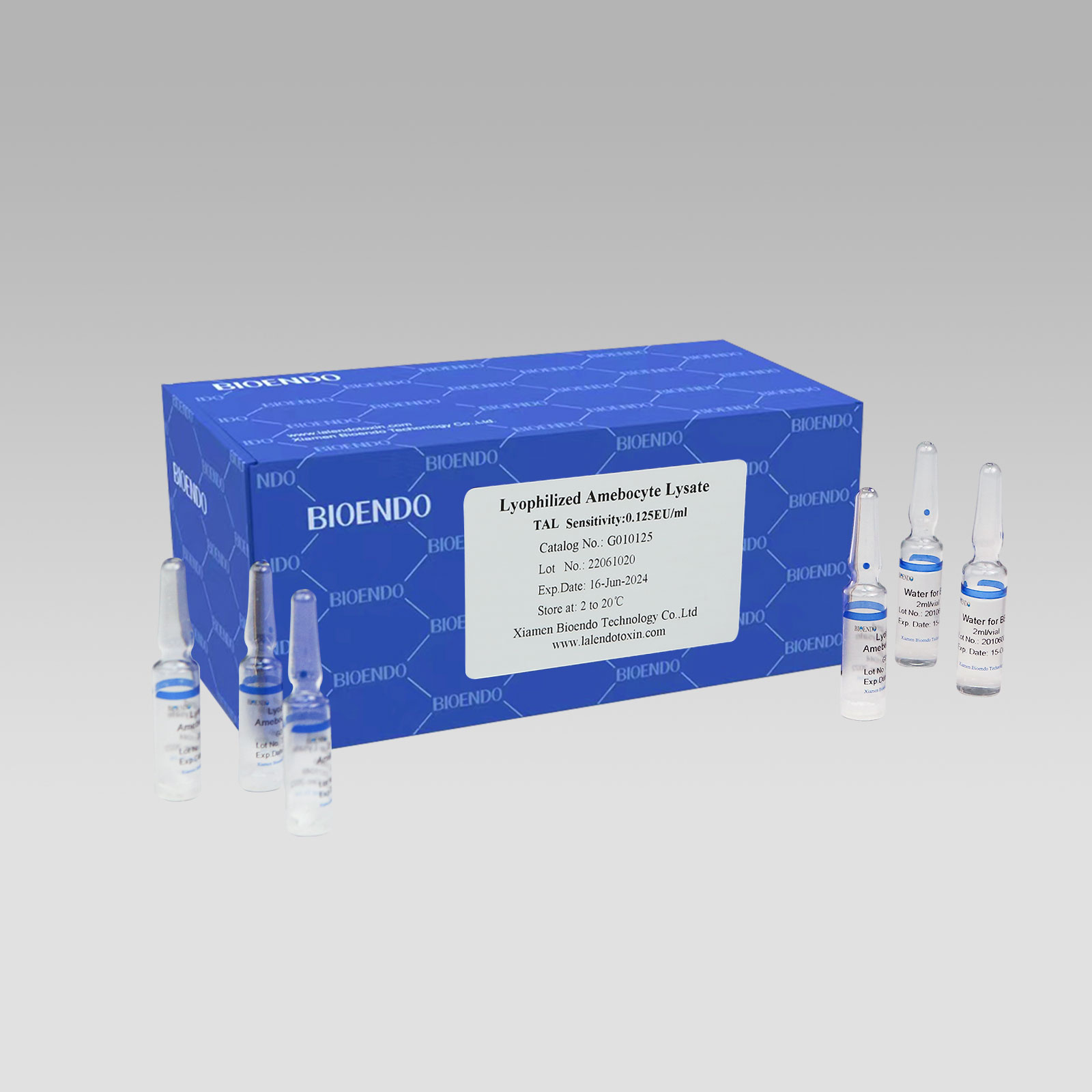
Gel Clot Lyophilized Amebocyte Lysate Single Test in Ampoule G01
Gel Clot Lyophilized Amebocyte Lysate Single Test Kit (Glass Ampoule G01)
The Gel Clot Lyophilised Amebocyte Lysate (LAL) Unius Testis in Ampoule est genus endotoxin testium tentandi, quod late in pharmaceuticis et medicinis artificiis industriis adhibitum est ad praesentiam endotoxorum in variis exemplis deprehendendi.
Hic sunt lineamenta quaedam huius testi endotoxini unius testi examinandi:
1. Sensibilitas: The Gel Clot Single LAL Test in Ampoule valde sensitivum est et endotoxin gradus deprehendere potest tam humilis quam 0.03 EU/mL.
2. Specificatio: endotoxin Imprimis, probatio endotoxins valde specifica est nec cum aliis substantiis in sample in cruce agere.
3. Commoditas: Unius examinis forma gel Clot Single LAL Test in Ampoule commodam et facilem ad usum facit, dum necessitatem reagentis et regulae curvae praeparandae removet.
4. Stabilitas: Forma lyophilisata de LAL reagentium optimam stabilitatem praebet, probationem ad cella temperiei protractum sine ullo eius effectu reponendam permittens.
5. Sumptus efficax: Unius examinis forma gel Clot LAL Singuli Testi in Ampoule plus efficax est quam alia genera endotoxini test pertentandi, sicut chromogenicum LAL primordium, in motu turbidimetric LAL primordium, extremum punctum chromogenicum LAL. primordium et recombinans factor C endotoxin test primordium.
The Gel Clot Single LAL Test in Ampoule est certa, sensitiva, et opportuna methodus ad endotoxinos in amplis exemplaribus detegenda, quod instrumentum essentiale ad salutem et qualitatem rerum pharmaceuticae et medicae machinae procurandae facit.
1. Product Information
Gel Clot Lyophilized Amebocyte Lysate Singulus Testin ampoule Continete endotoxin amebocyte Lysate specifica, quae includit inhibitorem beta-glucan in formula et non agere cum beta-glucan.Pro uno Testi nostro in ampoule vitreo, specimen addere potes ampoulis vitreis directe.Hoc modo non opus est ut Amebocyte Lysate in antecessum noviter restituas, et quot probationes singulis diebus ad vitandam vastitatem utantur, etiam haec est maxima differentia a endotoxin multi experimenti phialae.Endotoxinum liberum fistulae ad dilutionem Lyophilized CSE necessaria est.Operatio endotoxin detectionis usus Bioendo Gel Clot Lyophilised Amebocyte Lysate Singulus Testis in ampoule pharmacopoeiae nationali conformis.
2. Product Parameter
Gel concretum primordium unum test ampoule vitreum.
Sensus: 0.03EU/ml, 0.06EU/ml, 0.125EU/ml, 0.25 EU/ml, 0.5EU/ml.
3.Product Feature et Application
Unius gradus deprehensio endotoxin, facilis ad usum in experimento endotoxini bacterial.
Normalis aquae balnei vel sicci caloris incubator praesto est pro incubatione gel concreti methodus LAL reagens.
Apta forend-productum endotoxin temptationisante productum dimisit.
Operare endotoxin test primordium, rhoncus sensitivum normatum secundum pharmacopoeiam Sinarum criterium.
Nota:Lyophilised Amebocyte Lysate (LAL) gerens a Bioendo fabricatum factum est ex lysate amebocytarum (cellulae albae sanguinis) ex cancri solea.
| Catalogue No. | Sensitivum EU/ml | Descriptio | Ornamentum Contents |
| G010030 | 0.03 | Bioendo Gel Clot Endotoxin Test Kit, Single Test in Ampoule. | L probat per sarcina |
| G010060 | 0.06 | ||
| G010125 | 0.125 | ||
| G010250 | 0.25 | ||
| G010500 | 0.5 |
Sensus Lyophilised Amebocyte Lysate et potentia Imperii Latin Endotoxin tentantur contra USP Reference Standard Endotoxin.Lyophilised Amebocyte Lysate iodi rhoncus cum productum instructione veniunt, certificatorium Analyseos.
Quare maxime selecta gel concretum primordium G01 ornamentum;
1. Frugi maxime ac communiter probandi iodi deprehendendi endotoxin.
2. Singula experimenta in ampoule pro gradu contaminationis periculum minuunt.
3. Gel concretum primordium unum test ampoule vitreum non opus est sapiente microplate lectore.
4. Servans endotoxin liberum fistulam cum G01 serie utens ad probandos endotoxinos in processu operationis endotoxini experimenti tentandi.
Related products in endotoxin test primordium:
Aquae pro Bacterial Endotoxins Test (BET), TRW02, TRW50 vel TRW100;
Endotoxin gratis tubus vitreus (dilution tube), Pyrogenis apicibus liberae commenda T1310018, PT25096 vel PT100096 commenda
Pipettor,Suadeo PSB0220
Test Tube Rack;
Incubatio instrumenti
Suadeo calor arida Incubator TAL-M2
Vortex Mixter, Suadeo VXH.
Imperium Latin Endotoxin, CSE10A.